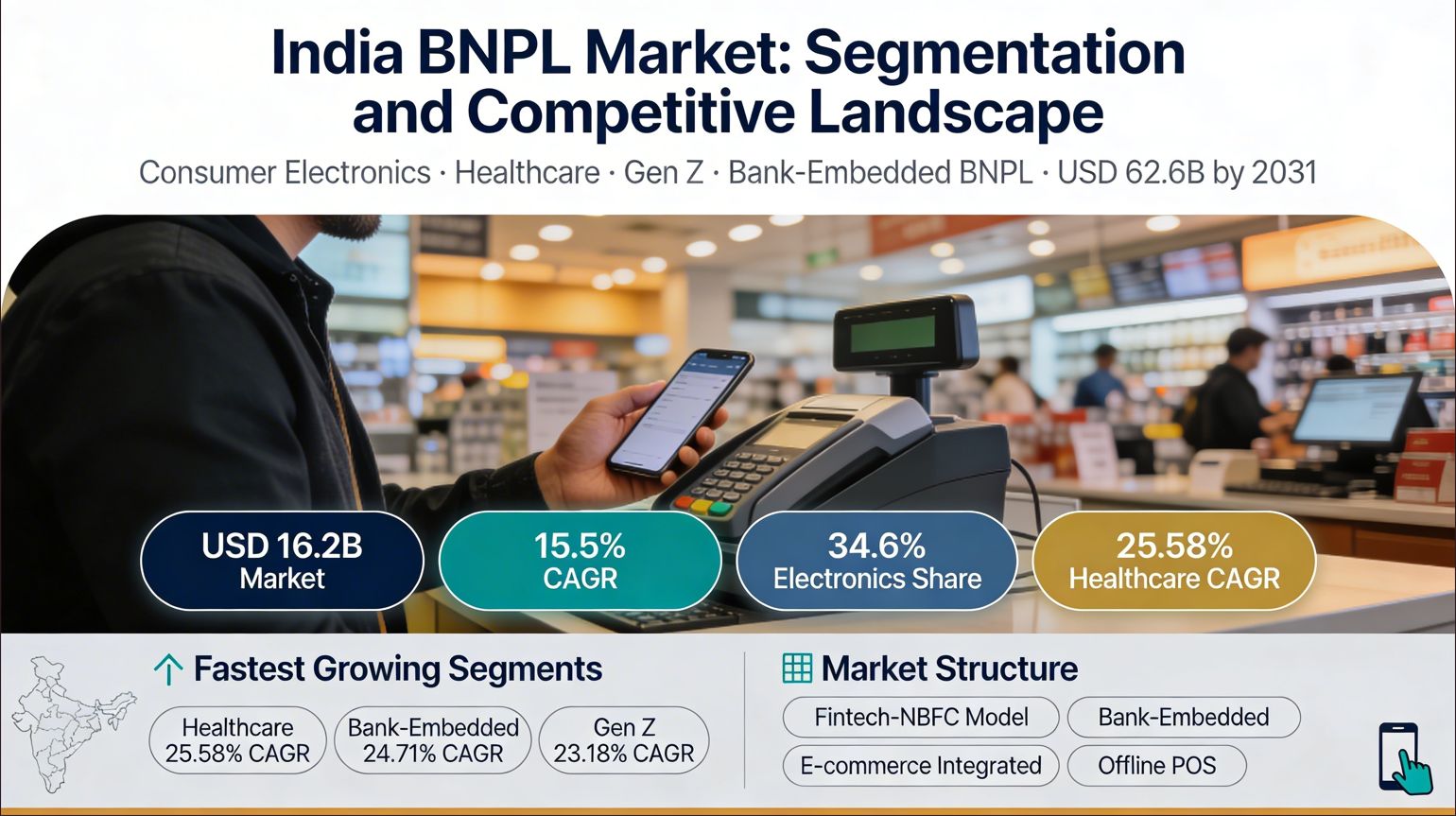
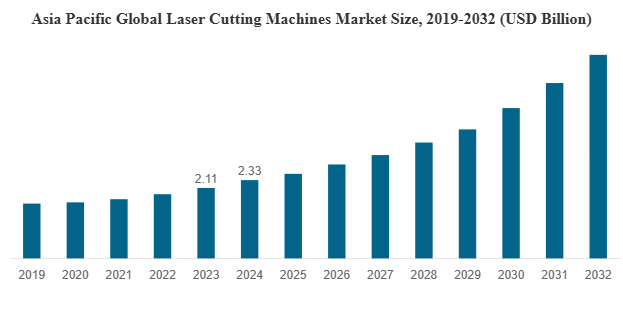
Cancer Diagnostics Market Size, Growth, Trends & Analysis 2025
👉 Best IPTV Services 2026 – 10,000+ Channels, 4K Quality – Start Free Trial Now
The global cancer diagnostics market was valued at USD 15.13 billion in 2023 and is expected to grow from USD 16.12 billion in 2024 to USD 31.00 billion by 2032, reflecting a compound annual growth rate (CAGR) of 8.5% during the forecast period. In 2023, North America led the market, holding a 35.89% share.
Cancer diagnostics, utilizing techniques like immunohistochemistry, in-situ hybridization, and others, play a crucial role in differentiating various cancer types and identifying disease subtypes. The rising global burden of cancer has been driving the demand for advanced diagnostic solutions that enable early detection. According to data from the World Health Organization (WHO), new cancer cases worldwide are projected to reach 35 million by 2050, marking a 77% increase from 20 million in 2022. Additionally, the growing emphasis of key industry players on research and development (R&D) for innovative diagnostic solutions has been propelling market growth. For example, in February 2023, F. Hoffmann-La Roche Ltd expanded its collaboration with Janssen Biotech Inc. to advance R&D efforts in companion diagnostics, including next-generation sequencing, digital pathology, polymerase chain reaction, and immunoassays.
Get a Free Sample Research PDF:
https://www.fortunebusinessinsights.com/enquiry/request-sample-pdf/cancer-diagnostics-market-110706
Market Segmentation:
By Product: The assay kits & reagents segment led the Cancer Diagnostics market in 2023 due to the rising number of cancer diagnoses and frequent product launches. For instance, Illumina, Inc. introduced the TruSight Oncology (TSO) test in May 2022 for molecular profiling. Meanwhile, the instruments segment is expected to grow steadily, driven by increasing global healthcare investments.
By Technique: Immunoassays dominated in 2023 due to their accuracy in detecting cancers like breast and prostate cancer. According to Breastcancer.org, around 310,720 women and 2,800 men were estimated to be diagnosed in the U.S. in 2024. The immunohistochemistry (IHC) segment is projected to grow fastest due to its precision and predictive capabilities.
By Type: Tissue biopsy held the largest share in 2023 due to its accuracy in initial diagnoses and molecular profiling, while liquid biopsy is expected to expand rapidly due to faster result turnaround—26.8 days quicker than tissue biopsy per an NCBI 2023 study.
By Cancer Type: Breast cancer led the Cancer Diagnostics market due to rising cases, with WHO reporting 2.31 million cases in 2022. However, lung cancer is expected to grow at the fastest CAGR due to increasing smoking addiction among youths.
By End-user: Clinical laboratories dominated in 2023 due to technological advancements and high test volumes, with 4 billion annual tests conducted in the U.S., per NCBI. Meanwhile, hospitals are expected to grow significantly due to patient preference for integrated diagnosis and treatment.
List Of Key Cancer Diagnostics Market Players Profiled In This Report:
• F. Hoffmann-La Roche Ltd (Switzerland)
• Thermo Fisher Scientific Inc. (U.S.)
• Abbott (U.S.)
• Illumina, Inc. (U.S.)
• GE Healthcare (U.S.)
• BD (U.S.)
• bioMérieux SA (France)
• Myriad Genetics, Inc (U.S.)
• Bio-Rad Laboratories, Inc. (U.S.)
• QIAGEN (Germany)
Cancer Diagnostics Market Driving Factors:
Rising Cancer Burden Driving Demand for Advanced Diagnostics
The global incidence of cancers, including prostate, breast, and colorectal cancer, has been increasing significantly. According to the American Cancer Society, the U.S. was projected to record 1,918,030 new cancer cases and 609,360 deaths in 2022. However, WHO data indicates that the actual number of new cases in the U.S. rose to 2,380,189, with an estimated 605,761 deaths. This rising disease burden underscores the need for early and effective diagnostics, which play a crucial role in timely treatment and reducing mortality rates. As a result, the demand for advanced cancer diagnostics continues to grow, driving global market expansion.
Growing Awareness and Screening Initiatives Boosting Market Growth
With increasing cancer prevalence, healthcare organizations and government bodies are intensifying awareness campaigns and screening programs to promote early detection.
For example, in August 2020, the World Health Assembly launched the Global Strategy for Cervical Cancer Elimination, emphasizing early screening and treatment. Governments worldwide are also implementing national screening programs, such as the ACCION initiative in the U.S., which promotes colorectal cancer (CRC) screening among uninsured Hispanic adults. Similarly, the U.K.'s Bowel Cancer Screening Programme (BCSP) conducts biennial screenings using fecal immunochemical tests (FIT). Additionally, research initiatives such as the Alliance of Glycologists for Cancer Research, Cancer Immunoprevention Network (CIP-Net), and Early Detection Research Network (EDRN) continue to advance cancer diagnostics. As governments and organizations prioritize awareness and early detection programs, the demand for innovative and effective cancer diagnostics is expected to rise, further propelling market growth.
Cancer Diagnostics Market Restraining Factors:
High Costs and Associated Risks May Hinder Market Growth
While the rising prevalence of cancer and increased awareness of early diagnosis are driving demand for advanced diagnostics, certain challenges are limiting market expansion. Colonoscopy, widely regarded as the gold standard for colorectal cancer screening in countries like the U.S., carries risks such as bleeding, perforation, and device-associated infections, which can deter adoption. Additionally, the high cost of diagnostic tools poses a significant barrier. For example, PCR machines range from USD 3,000 to USD 30,000 and require regular maintenance, making them less accessible, particularly in emerging economies. Many healthcare facilities struggle to expand diagnostic services due to these financial constraints, impacting the effectiveness of cancer detection. As a result, the high costs and potential risks associated with cancer diagnostics may restrict broader market growth despite increasing demand.
Key Industry Developments:
• August 2024: Illumina, Inc. expanded the oncology menu for its NovaSeq X Series diagnostic equipment, strengthening its cancer diagnostics portfolio.
• April 2024: F. Hoffmann-La Roche Ltd received CE Mark approval for the VENTANA HER2 (4B5) Rabbit Monoclonal Primary Antibody RxDx, designed to detect metastatic breast cancer patients with low HER2 expression.