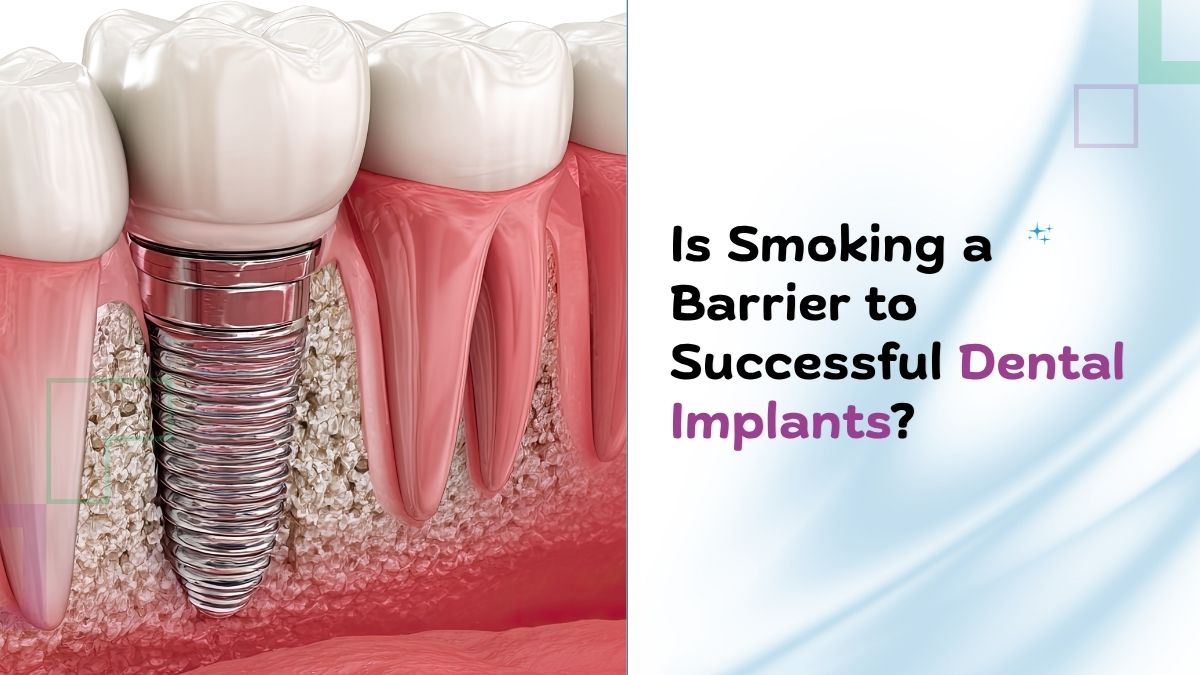
Miradri Treatment Explained: Benefits, Procedure, Recovery, and Patient Experiences
FREE SEO Topical Map Generator: Find Your Next Content Ideas
Intent: Informational
Miradri treatment is an increasingly discussed option in ophthalmology and outpatient eye care for targeted pupil or anterior segment management. This guide explains what Miradri treatment typically involves, who may be a candidate, the likely benefits and risks, and what patients report during recovery.
- Miradri treatment: an outpatient ocular intervention used to manage pupil behavior or anterior segment symptoms (term used here as a clinical treatment category).
- Benefits: faster symptom relief, potentially lower systemic exposure than oral medications, targeted effect.
- Process: pre-evaluation, in-office procedure (topical agents or device-assisted), short observation, brief recovery period.
- Common concerns: temporary blurred vision, light sensitivity, and short-term discomfort.
- Next step: consult an eye-care specialist for diagnosis, candidacy assessment, and informed consent.
Miradri treatment: benefits, process, and expectations
Miradri treatment often aims to achieve a focused therapeutic effect in the eye using topical agents or a minor in-office procedure; benefits can include faster onset of action, targeted local effect, and the ability to perform the intervention without general anesthesia. This section outlines typical outcomes, how the process looks, and related terminology such as ocular pharmacology, pupil modulation, and anterior segment therapy.
What Miradri treatment is and who it helps
The term covers interventions designed to alter pupil size or anterior segment behavior for symptomatic relief or to improve visualization during diagnostic or minor procedures. Candidates are generally adults evaluated for specific ocular indications by an ophthalmologist or optometrist. Contraindications may include allergy to topical agents, certain cardiac conditions, or uncontrolled systemic illnesses—professional evaluation is required.
Typical benefits reported
- Targeted symptom control with minimal systemic exposure.
- Quick in-office application and fast clinical effect.
- Reduced need for more invasive procedures in selected cases.
Procedure steps and what to expect before, during, and after
Pre-procedure evaluation
Standard pre-procedure steps include a medical history review, medication reconciliation, and baseline vision testing. Patients should disclose prescription medications, over-the-counter products, and any cardiovascular history.
During the Miradri procedure
The intervention typically takes 10–30 minutes in a clinic setting. Local topical agents or a device are applied; comfort measures and brief monitoring follow. No general anesthesia is usually needed, and driving restrictions may apply immediately after.
Miradri procedure recovery
Recovery timelines vary depending on the exact technique. Many patients notice effects that wear off in hours to a few days. Common short-term experiences include blurred near vision, light sensitivity, or mild irritation. If symptoms persist beyond the expected window, follow-up evaluation is recommended.
Miradri side effects: what patients report
Reported side effects are commonly transient and local: temporary blurred vision, photophobia, mild stinging on application, or short-lived headache. Serious adverse effects are uncommon but require urgent assessment. Medication interactions and allergy history are important to review before treatment.
Practical framework and checklist for consultations
Use the MIRADRI Checklist to structure patient consultations and shared decision-making:
- Mechanism — Explain how the intervention works and what it targets.
- Indication — Confirm the specific reason for the treatment and alternatives.
- Risks — Review common and rare side effects and interactions.
- Aftercare — Detail recovery steps, follow-up timing, and activity limits.
- Data — Share expected outcomes and evidence basis for the procedure.
- Recovery — Describe the timeline and signs that warrant a call back.
- Informed consent — Ensure questions are answered and consent is documented.
Real-world example
Scenario: A 62-year-old patient with intermittent symptomatic photophobia and difficulty with near tasks visits an eye clinic. After evaluation and medication review, an in-office Miradri application is performed to temporarily modulate pupil response. The patient experiences immediate improvement in glare for several hours, with mild blurred near vision that resolves the same day. A follow-up visit at one week confirms symptom reduction and no adverse events.
Practical tips for patients and clinicians
- Bring a list of all current medications and health conditions to the consultation to avoid interactions.
- Arrange a ride or limit driving for the day of treatment if vision may be temporarily impaired.
- Follow aftercare instructions closely—use prescribed drops, avoid rubbing eyes, and report persistent pain or vision loss immediately.
- Ask for written information about expected outcomes and who to contact for urgent questions.
Trade-offs and common mistakes
Trade-offs to consider
Choosing Miradri treatment can offer rapid, targeted relief but may require repeat applications for sustained benefit. Compared with systemic medications, local interventions often reduce systemic side effects but might be less durable.
Common mistakes
- Skipping a full medication review before treatment, which may raise interaction risks.
- Underestimating the short-term visual effects—patients should be warned about temporary blur and light sensitivity.
- Failing to provide clear follow-up instructions, leading to unnecessary emergency visits for expected, transient symptoms.
Core cluster questions for related articles and internal linking
- How does Miradri treatment compare with standard topical eye medications?
- What are the long-term effects and follow-up needs after Miradri-style interventions?
- Which patients are poor candidates for pupil-modulating in-office procedures?
- How should clinicians document informed consent for short outpatient ocular procedures?
- What aftercare practices improve patient comfort and recovery after eye interventions?
Related entities and synonyms: ocular therapy, pupil modulation, anterior segment treatment, topical ophthalmic agents, outpatient eye procedure, ophthalmology practice, patient-reported outcomes.
For regulatory context on medical device and drug safety processes, see the U.S. Food and Drug Administration guidance on medical devices and approvals: FDA Medical Devices.
Practical next steps
To decide whether Miradri treatment is appropriate, schedule a diagnostic evaluation with a licensed eye-care professional, request a copy of the MIRADRI Checklist during the visit, and clarify expectations about benefits and recovery time.
Patient experiences and evidence notes
Patient reports often emphasize quick symptom relief and short recovery. Clinical evidence should be reviewed through peer-reviewed ophthalmology literature and specialist guidance; request that the treating clinician share references relevant to the specific technique being offered.
What is Miradri treatment and who is a candidate?
Miradri treatment generally refers to a targeted ocular intervention delivered in an outpatient setting. Candidates are selected after an eye exam and medical history review; decisions are individualized based on indication and risk factors.
How long does Miradri procedure recovery typically take?
Recovery can range from a few hours to several days, depending on the approach used. Most patients return to normal activities quickly but may be advised to avoid driving until vision clears.
What are common Miradri side effects and how are they managed?
Common effects include temporary blurred vision and light sensitivity. Management usually involves rest, sunglasses, and symptomatic topical drops when recommended. Persistent or severe symptoms require prompt follow-up.
How to prepare for a Miradri treatment consultation?
Bring medication and allergy lists, a recent health summary, and prepare questions about alternatives, expected outcomes, and follow-up care.
When to seek emergency care after Miradri treatment?
Seek urgent assessment for sudden vision loss, severe pain, signs of infection (increasing redness, discharge, fever), or any new neurologic symptoms after treatment.