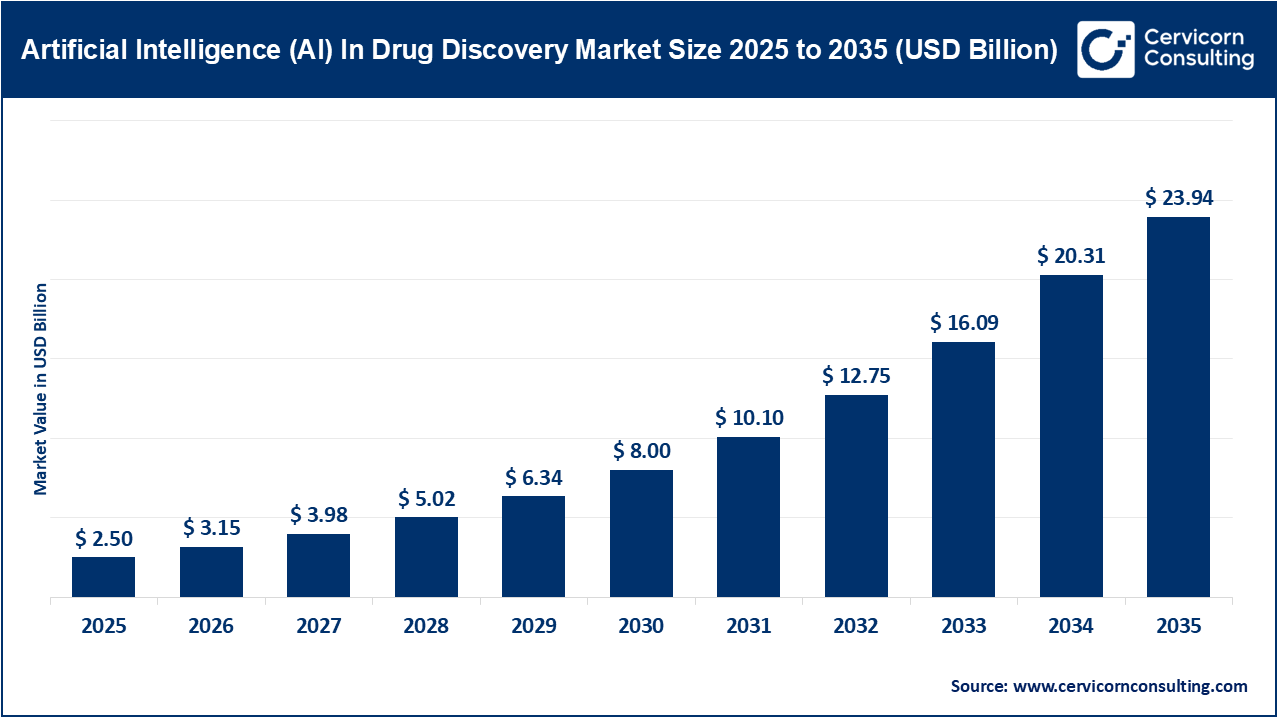
Oxycodone 30 mg: Uses, Dosage Guidelines, Risks, and Safe-Use Checklist
Detected intent: Informational
Oxycodone 30 mg is a prescription opioid formulation used for moderate to severe pain when alternative treatments are inadequate. This article explains how oxycodone 30 mg works, when it may be appropriate, key safety considerations, and practical guidance for safer use.
Oxycodone 30 mg can provide strong analgesia but carries significant risks including respiratory depression, dependence, and overdose. Use only under clinician supervision, follow dosing and monitoring recommendations, and apply a harm-reduction checklist before prescribing or taking it. See the SAFER Pain Checklist and practical tips below.
Oxycodone 30 mg: Uses, dosage, and risks
Oxycodone 30 mg is typically reserved for patients with severe acute pain (for example, postoperative pain) or certain chronic pain conditions where lower-strength opioids or non-opioid analgesics are ineffective or inappropriate. Because 30 mg is a high single dose for many patients, clinicians evaluate prior opioid tolerance, renal and hepatic function, and concurrent medications before prescribing.
How oxycodone works and key pharmacology
Oxycodone is a semi-synthetic opioid agonist that binds mu-opioid receptors in the central nervous system, reducing pain perception and altering pain response. Onset, peak effect, and duration depend on formulation (immediate-release vs extended-release), patient metabolism, and interactions with other central nervous system depressants like benzodiazepines or alcohol. Common pharmacologic concerns include respiratory depression, constipation, sedation, tolerance, and physical dependence.
Who may be considered for oxycodone 30 mg?
Appropriate candidates generally include patients with severe pain requiring potent opioid therapy and demonstrable tolerance to opioids—often those already stabilized on lower doses. Oxycodone 30 mg is not suitable for opioid-naive patients without a careful titration plan. Special caution is required for older adults, people with sleep-disordered breathing, and those with hepatic or renal impairment.
Dosage guidelines and conversion considerations
Converting from another opioid to oxycodone 30 mg requires validated equianalgesic tables and clinical judgment. Tapering or dose adjustments should account for incomplete cross-tolerance. For safety, clinicians commonly reduce calculated equianalgesic doses by 25–50% and monitor closely. Hospital protocols and pain-management guidelines from professional societies provide specific conversion charts and monitoring intervals.
SAFER Pain Checklist (a practical framework)
Use the SAFER Pain Checklist before prescribing or taking oxycodone 30 mg. This five-point checklist helps reduce avoidable harm:
- Screen: Confirm pain diagnosis, opioid tolerance, and contraindications.
- Alternatives: Document trials of non-opioid and non-pharmacologic treatments.
- Formulation & Dose: Choose immediate vs extended-release appropriately and verify dose conversions.
- Education: Provide patient education on risks, storage, and overdose response (including naloxone availability).
- Response plan: Set goals, follow-up schedule, and criteria for discontinuation or tapering.
Practical example: postoperative pain scenario
A 58-year-old patient undergoes major abdominal surgery and reports severe pain despite scheduled acetaminophen and NSAIDs. The surgeon prescribes short-term oxycodone 30 mg immediate-release for breakthrough pain only, with clear instructions: maximum frequency, expected duration (48–72 hours), monitoring for sedation or breathing difficulty, and return visit for reassessment. The discharge paperwork includes naloxone information and instructions to avoid alcohol and other sedatives. This example shows an appropriate brief, monitored use rather than long-term prescribing.
Practical tips for safer use
- Confirm opioid tolerance before prescribing oxycodone 30 mg; avoid using it in opioid-naive patients without a stepwise titration plan.
- Check for drug interactions (especially benzodiazepines, gabapentinoids, and certain antidepressants) that increase sedation or respiratory risk.
- Provide written instructions on safe storage and disposal to prevent diversion or accidental ingestion.
- Discuss naloxone with patients at higher risk for overdose and provide education on signs of overdose and emergency steps.
- Schedule an early follow-up to reassess pain control, side effects, and functional goals; document shared decision-making.
Trade-offs and common mistakes
Prescribing oxycodone 30 mg can offer rapid and effective pain relief, but trade-offs include a higher risk of respiratory depression, increased potential for misuse or dependence, and more pronounced side effects compared with lower-dose options. Common mistakes include:
- Starting high in opioid-naive patients without titration.
- Failing to check for sedative co-medications or sleep apnea.
- Not providing clear take-home instructions, naloxone info, or follow-up plans.
- Skipping documentation of alternative therapies attempted before escalating to high-dose opioids.
Regulatory and safety references
Regulatory bodies emphasize caution with opioid prescribing and recommend patient education, risk assessment, and access to reversal agents. For consolidated federal guidance on opioid risks, labeling, and safety communications, see this resource: FDA opioid analgesics information.
Core cluster questions
- When is oxycodone 30 mg appropriate for acute vs chronic pain?
- How to convert other opioids to oxycodone 30 mg safely?
- What are the early warning signs of oxycodone overdose and how should naloxone be used?
- How does renal or hepatic impairment change oxycodone dosing?
- What non-opioid strategies should be tried before prescribing high-dose oxycodone?
Monitoring and discontinuation
Monitoring should include assessment of pain relief, functional improvement, sedation scales, respiratory status, and signs of misuse. Tapering is indicated if goals are unmet, side effects are unacceptable, or evidence of misuse emerges. Gradual dose reductions (for example, 10%–25% per week depending on duration and dependence) minimize withdrawal, but individualized plans are essential.
When to seek urgent attention
Seek immediate medical help for shallow or stopped breathing, severe drowsiness with inability to wake, blue lips or nails, or signs of acute allergic reaction. Emergency services should be contacted promptly in suspected overdose situations.
Conclusion
Oxycodone 30 mg can be an effective option for severe pain but requires careful candidate selection, dose planning, patient education, and monitoring. Applying a structured checklist like SAFER and following best-practice prescribing principles reduces risks while preserving access to necessary analgesia.
Frequently asked questions
Is oxycodone 30 mg appropriate for opioid-naive patients?
No. Oxycodone 30 mg is generally too high for opioid-naive individuals. A stepwise titration using lower-dose immediate-release formulations is safer, and clinicians should evaluate tolerance before prescribing higher-dose tablets.
How long should a typical short-term oxycodone 30 mg prescription last?
Short-term use is typically limited to a few days to a week for acute postoperative pain, with frequent reassessment. Longer prescriptions require clear goals, monitoring plans, and documentation of alternatives tried.
What are the most common side effects of oxycodone 30 mg?
Common side effects include constipation, nausea, drowsiness, dizziness, and respiratory depression in higher doses; proactive management of constipation and monitoring for sedation are standard practices.
Can oxycodone 30 mg interact with other medications?
Yes. Significant interactions occur with benzodiazepines, alcohol, sedative-hypnotics, and certain CYP3A4 inhibitors or inducers that alter oxycodone metabolism, increasing risk of sedation or withdrawal symptoms.
How should oxycodone 30 mg be stored and disposed of?
Store in a locked location out of reach of children and others, and follow local drug-take-back programs or pharmacy disposal recommendations to prevent diversion and accidental ingestion.