Dubai Glutathione Injection Safety Guide: Do’s and Don’ts
Get a free topical map and start building content authority today.
Glutathione injections in Dubai are offered in a variety of clinical settings for perceived antioxidant or cosmetic benefits. This guide explains practical do's and don'ts to help residents and visitors make informed choices, focus on safety, and understand regulatory considerations when considering injectable glutathione.
- Verify clinic licensure and practitioner qualifications before any injection.
- Discuss medical history, medications, and allergies with a qualified clinician.
- Understand the differences between intravenous and intramuscular routes and possible side effects.
- Report adverse events to local health authorities and keep product batch information.
Glutathione Injections in Dubai: Key Do’s and Don’ts
Do: Check regulation and provider credentials
Confirm that the clinic and practitioner are licensed by the relevant UAE health authority and by local emirate regulators. Request to see professional qualifications, clinic registration, and current licenses. For regulatory guidance and contact details in the UAE, consult the Ministry of Health and Prevention (MOHAP) website: https://www.mohap.gov.ae.
Don’t: Proceed based on marketing or social media alone
Avoid choosing a provider solely on social-media claims, before-and-after photos, or promotions. Marketing may not reflect safety practices, sterile technique, or the evidence base for benefits. Look for transparent informed-consent documents, written treatment plans, and clear pricing that includes follow-up care.
Do: Have a clinical consultation first
Request a pre-procedure consultation to review medical history, current medications, allergies, and any prior reactions to injections. Discuss pregnancy, breastfeeding, chronic conditions, and any genetic enzyme deficiencies that may be relevant. Clinicians should document the discussion and obtain informed consent before administering injections.
Don’t: Hide medications or supplements
Disclose all prescription drugs, over-the-counter medications, and dietary supplements. Potential interactions and effects on liver function, blood counts, or antioxidant systems are part of clinical assessment. A qualified clinician will assess suitability and advise on temporary discontinuation of certain agents if needed.
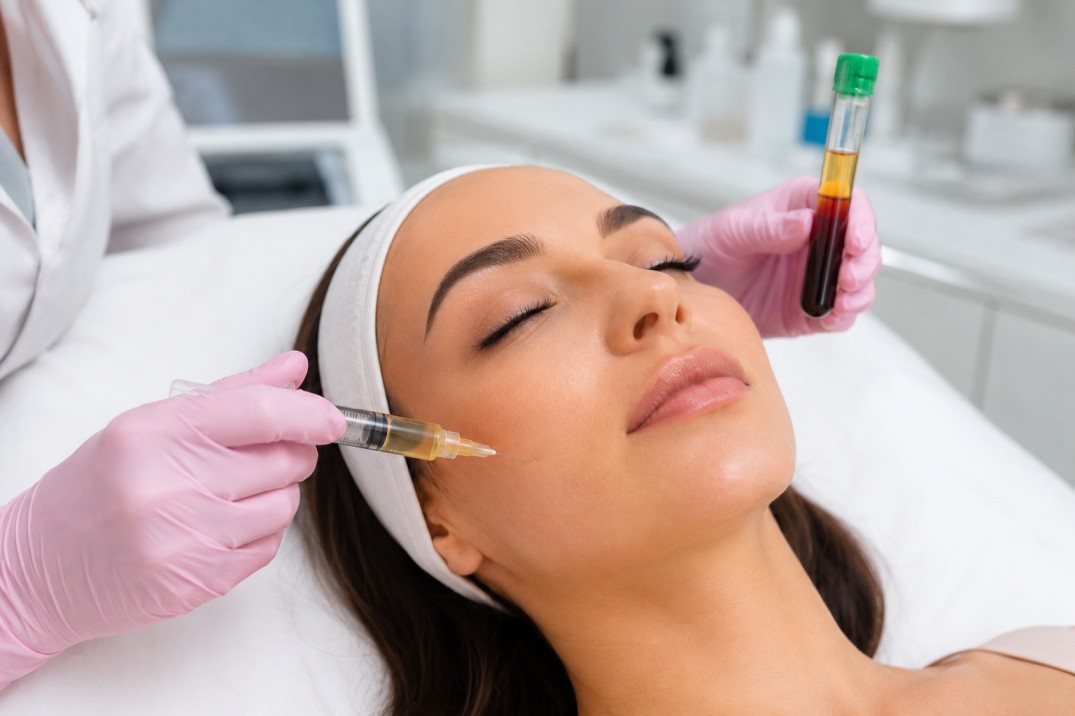
Do: Understand administration methods and product quality
Learn whether the clinic offers intravenous (IV), intramuscular (IM), or other routes. Proper sterile technique, single-use syringes, and clear documentation of the product name, manufacturer, lot number, and expiry date are important quality-control measures. Many injections are prepared by licensed compounding pharmacies or supplied as manufactured sterile products—ask how the product is sourced and stored.
Don’t: Accept unclear dosing or unlabelled vials
Decline treatment if the provider cannot show the vial label, batch information, or if dosing instructions are vague. Excessive or unstandardized dosing regimens increase uncertainty about effectiveness and safety. If a clinic suggests unusually high frequencies or combines injections with unproven therapies without explanation, seek a second opinion.
Do: Expect monitoring and immediate response for reactions
During and after administration, the clinic should monitor vital signs and be prepared to manage allergic reactions, local injection-site problems, or systemic symptoms. Know where the nearest emergency services are located and confirm that the facility has basic resuscitation equipment and trained staff.
Don’t: Ignore side effects or skip follow-up
Report swelling, persistent pain, high fever, trouble breathing, unusual bruising, jaundice, or other concerning symptoms right away. Keep records of treatment dates and product details for any adverse-event reports.
Safety, evidence and reporting
Understand the evidence and limitations
Scientific literature on injectable glutathione covers biochemical effects and small clinical studies for specific indications, but high-quality evidence for cosmetic claims such as skin-lightening or long-term health benefits is limited. National and international health organizations encourage transparent reporting of adverse events and careful evaluation of risk versus benefit before elective aesthetic procedures.
Report adverse events and keep documentation
If an adverse reaction occurs, report it to the treating provider and to local health authorities. In the UAE, the Ministry of Health and Prevention collects safety reports. Retain product packaging, batch numbers, and the clinic’s documentation to facilitate investigation.
Consider alternatives and non-invasive options
Discuss evidence-based alternatives or non-invasive approaches for the intended outcome. A licensed clinician can outline risks, expected outcomes, and whether a trial of other interventions is appropriate before injections.
Common practical checklist before booking
- Verify clinic and practitioner licensure and inquire about training in injectable therapeutics.
- Request product details: name, manufacturer, lot number, expiry date, and source.
- Ask about sterile technique, single-use disposables, and emergency preparedness.
- Obtain written informed consent, a treatment plan, and instructions for aftercare.
- Know how to contact the clinic for post-procedure concerns and how to report adverse events to health authorities.
Frequently asked questions
Are Glutathione injections in Dubai safe?
Safety depends on the product, dose, route, and the provider’s practices. When given by a licensed clinician using properly sourced, sterile products and appropriate monitoring, risks may be reduced. However, as with any injectable treatment, there are potential side effects, and evidence for many cosmetic uses is limited. Consult a licensed health professional to discuss individual risks.
How are injections administered and what are common side effects?
Glutathione can be administered intravenously or intramuscularly. Common side effects include local pain at the injection site, redness, or transient fatigue. More significant reactions—such as allergic responses, injection-site infections, or systemic symptoms—require prompt medical evaluation.
How should a clinic verify product quality?
Products should be labelled with manufacturer information, batch number, and expiry date. Ask whether vials are single-use, how they are stored, and whether they are sourced from licensed manufacturers or compounding pharmacies that meet regulatory standards.
What steps should be taken if an adverse reaction occurs?
Seek immediate medical care for severe symptoms. Report the event to the treating clinic and to local health authorities, providing product and batch information if available. Retain documentation and follow-up as advised by a licensed clinician.
How to choose a clinic for aesthetic injections?
Choose a clinic with clear licensure, documented practitioner training, evidence of sterile technique, transparent sourcing of products, and a documented process for informed consent and follow-up care. Favor facilities that can explain risks and alternatives without pressure to purchase immediate or repeated treatments.
Can pregnant or breastfeeding people receive glutathione injections?
Pregnant or breastfeeding people should discuss potential risks with a licensed clinician. Many injectable products lack robust safety data in pregnancy and lactation, so individual assessment is important.
Who regulates injectable therapies in the UAE?
Regulation is overseen by national and emirate health authorities. For general information and contact details, see the UAE Ministry of Health and Prevention: https://www.mohap.gov.ae.
Note: This article provides general information and does not replace individualized clinical advice. For personal medical decisions, consult a licensed healthcare professional or regulator in the UAE.