Skin Whitening Injections: A Practical Safety Guide and Decision Checklist
Get a free topical map and start building content authority today.
Detected intent: Informational
Skin whitening injections are an increasingly discussed cosmetic option. This guide explains what skin whitening injections are, how they claim to work, documented risks, and a practical decision checklist for anyone considering treatment. The phrase skin whitening injections appears here to anchor the topic and guide further reading.
- Skin whitening injections commonly use glutathione or vitamins; supporting evidence for long-term lightening is limited.
- Risks include allergic reactions, infection, organ toxicity, and unregulated product quality.
- Use a safety checklist (SAFE framework) and consult licensed dermatology or medical providers before treatment.
Skin whitening injections: what they are and how they work
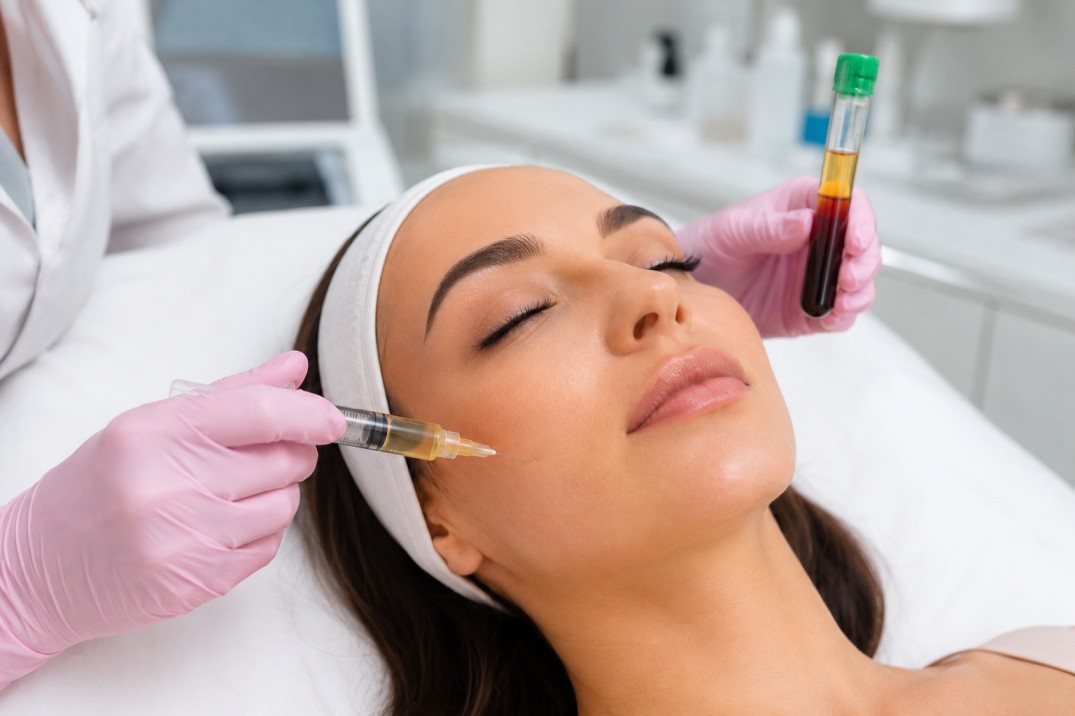
Skin whitening injections are injectable preparations marketed to reduce melanin production or alter skin tone. The most commonly discussed agent is glutathione, an amino-acid antioxidant administered intravenously (IV) or intramuscularly; other formulations mix vitamins, corticosteroids, or unverified compounds. Claims typically center on reduced melanin synthesis, antioxidant activity, or transient flushing effects. Clinical evidence for consistent, long-term skin lightening is limited, and many products and practices remain off-label or unapproved.
Types, mechanisms, and common compounds
Glutathione-based injections
Glutathione is proposed to shift melanin synthesis toward lighter pheomelanin and reduce oxidative stress. Research results are mixed; some small studies show short-term changes, while higher-quality trials are lacking. Routes include IV infusion, intramuscular injection, or oral supplements with variable bioavailability.
Vitamin and co-factor blends
Some clinics offer combinations of vitamins (B-complex, vitamin C), nicotinamide, or amino acids. These may improve skin appearance by supporting general skin health but are not established as reliable whitening agents.
Off-label or unregulated compounds
Corticosteroids and other agents may be used illicitly for bleaching; these carry clear systemic risks such as adrenal suppression and skin thinning. Product quality and labeling are common concerns when treatments are not regulated.
Safety, regulation, and documented risks
Safety issues are central. Adverse events can range from mild injection-site reactions to severe systemic outcomes like anaphylaxis, acute kidney injury, or hepatic strain. Unregulated products increase risk of contamination or incorrect dosing. Regulatory bodies vary by country; in the United States the U.S. Food and Drug Administration (FDA) provides general guidance on cosmetic injectables and product safety, and products without approval or proper manufacturing control are higher risk (U.S. Food and Drug Administration — Cosmetics).
Common documented risks
- Allergic or hypersensitivity reactions, potentially severe
- Infection at injection sites, including bacterial or viral transmission
- Systemic organ effects (liver, kidney) from high or repeated dosing
- Unpredictable pigmentary changes, paradoxical hyperpigmentation
- Psychological or body-image consequences when expectations are unmet
SAFE framework: a decision checklist before any injection
Use the named SAFE framework before proceeding. SAFE is a concise, practitioner-oriented checklist to evaluate suitability and reduce risk.
- Source verification: Confirm product identity, batch testing, and manufacturer credentials.
- Assessment: Medical history, allergy check, liver/kidney baseline labs, and realistic expectation setting.
- Facility and provider credentials: Licensed clinic, sterile technique, emergency protocols, and trained clinician present.
- Education and consent: Written informed consent, explanation of off-label use, alternatives, and documented follow-up plan.
Real-world example
Scenario: A 34-year-old with darker phototype skin requests IV glutathione for cosmetic lightening. Applying SAFE: verify the product is pharmaceutical-grade, obtain baseline liver and kidney tests, confirm no pregnancy, review evidence limits and alternatives (topical agents, sun protection), ensure treatment is in a licensed clinic, and document consent with plan for scheduled follow-up and lab monitoring.
Practical tips for safer decisions
- Ask for product documentation: batch numbers, certificates, and manufacturer contact details before treatment.
- Choose a licensed medical facility with trained staff and emergency preparedness; avoid informal or home-based injections.
- Request baseline blood tests (liver enzymes, renal function) before repeated IV or high-dose treatments.
- Prioritize evidence-based options: topical hydroquinone alternatives, retinoids, chemical peels, and laser therapies have clearer risk profiles and published data.
- Allow at least one week for post-injection monitoring and understand how to report adverse events.
Trade-offs and common mistakes
Trade-offs:
- Potential quick results versus limited long-term evidence and safety unknowns.
- Lower cost providers versus higher risk from unregulated products or poor sterile technique.
- Desire for uniform tone versus risk of uneven or paradoxical pigmentation.
Common mistakes:
- Assuming 'natural' or 'vitamin' labels guarantee safety.
- Skipping medical assessment or baseline labs for repeated IV use.
- Not documenting informed consent about off-label or unapproved use.
When to seek professional care
Seek urgent care if symptoms of severe allergic reaction occur (difficulty breathing, swelling, widespread rash), signs of infection develop (fever, increasing redness, pus), or any new systemic symptoms like jaundice, dark urine, or decreased urine output. Report any unexpected pigment changes to a qualified dermatologist for evaluation.
Core cluster questions (for related articles or internal linking)
- How effective are glutathione injections for skin lightening?
- What are the medical risks of intravenous skin lightening treatments?
- Which topical alternatives to injections are evidence-based for skin lightening?
- How to verify the quality and safety of injectable cosmetic products?
- What medical tests are recommended before cosmetic injectables?
Final considerations
Skin whitening injections are a contested and imperfectly regulated area. Careful evaluation, use of the SAFE framework, reliance on licensed providers, and realistic expectations reduce risk. For many people, proven topical treatments and sun protection offer safer, evidence-backed improvement for uneven tone.
Are skin whitening injections safe?
Safety depends on the compound, provider, and oversight; documented risks exist. Follow the SAFE checklist and consult a licensed medical provider for personalized assessment.
What is glutathione and does it whiten skin?
Glutathione is an antioxidant involved in cellular processes. Some small studies report short-term skin tone changes, but evidence for reliable, long-term whitening is limited and not uniformly supported by high-quality trials.
How to verify injectable product quality?
Request batch documentation, certificates of analysis, and supplier information. Prefer products from established pharmaceutical manufacturers and clinics that maintain cold-chain and storage records.
What alternatives exist to injections for lighter skin tone?
Topical agents (hydroquinone, azelaic acid, retinoids), chemical peels, and certain laser or light therapies have more robust clinical data and predictable safety profiles when performed by qualified professionals.
How many treatments are typical and is maintenance required?
Protocols vary widely. Many providers recommend multiple sessions with maintenance dosing; consider that repeated exposure increases cumulative risk and requires medical monitoring.